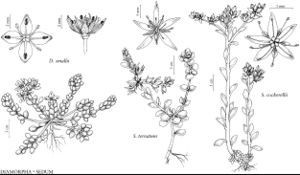
Diamorpha smallii
in N. L. Britton et al., N. Amer. Fl. 22: 56. 1905,.
Herbs mostly bright red, some green, 2–10 cm diam., roots white, fibrous. Stems simple beneath cyme or often with 1–several branches from crowded rosette nodes; hypocotyl 0.2–3 cm × 0.5–2 mm. Leaves: petiole slender; blades of winter rosette spatulate, to 10 × 2 mm; cauline leaves spreading or ascending, some transitional, most sessile; blade 1–3 mm wide, ca. 1 mm thick, apex obtuse. Cymes 2–200-flowered, sometimes flowers solitary, branched; branches each 1–8-flowered. Pedicels slender, 1–6 mm, thickened apically. Flowers open 3–5 days, with faint sweet odor; sepals deltate, 0.5–1 mm; petals convolute, boatshaped, cucullate, 1.5–3.5 × 1–1.5 mm, much longer than sepals, apex obtuse; filaments 1–2 mm, shorter than petals; anthers dark red, 0.2–0.4 mm; pistils connate about conic receptacle, white becoming red, lanceolate, 1–3.5 mm; styles 0.5–0.8 mm. Capsules brown, 3–8 mm diam., valves rhombic-ovate, 2 × 0.7 mm, apex acute. Seeds 2–12 per cell. 2n = 18.
Phenology: Flowering spring.
Habitat: Thin gravel and sand of vernal pools on flat granitic and sandstone rocks, sandy flats
Elevation: 100-600 m
Distribution
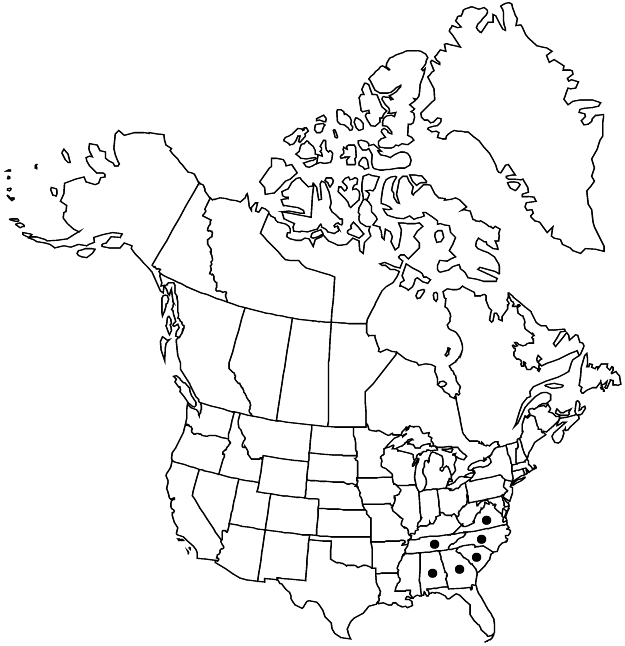
Ala., Ga., N.C., S.C., Tenn., Va.
Discussion
The correct name for this species has been much debated because of an early confusion with Sedum pusillum (R. T. Clausen 1975; R. L. Wilbur 1964, 1977). Wilbur argued that the long-used name Diamorpha cymosa (Nuttall) Britton is illegitimate.
Diamorpha smallii has been the subject of a remarkable number of studies, including some unpublished theses cited by various authors. D. N. Wiggs and R. B. Platt (1962) and J. F. McCormick and Platt (1964) studied the life history and ecology. Seeds germinate with autumn rains and warm temperatures, and the plants remain semidormant as rosettes during dry times until spring, when they flower, set seed, and die. Seeds seem to be distributed by water.
R. McVaugh (1943), C. Keever et al. (1951), and M. P. Burbanck and R. B. Platt (1964) studied plant succession in depressions of the flat rocks of the Piedmont region. Diamorpha is a primary invader in this harsh environment, partly because of its tolerance for submergence after the rains and for drought and heat later on. Seeds reaching other areas may germinate but the seedlings then are shaded out by other plants.
Although bees also carry the pollen, the main pollinators of Diamorpha seem to be ants of two common species, Formica schaufussi Mayr and F. subsericea Say (R. Wyatt 1981; Wyatt and A. Stoneburner 1981). The plants appear to be self-incompatible; there was no seed set from self-pollination or in greenhouse plants with no pollinators.
J. F. McCormick and R. B. Platt (1964) reinforced the original chromosome count with apparently 80 or more counts on plants from 16 populations, always finding n = 9. That is the same number as for the Californian Sedella, likewise annuals and with an unusual but totally different type of fruit.
Selected References
None.