Crataegus rivulopugnensis
J. Bot. Res. Inst. Texas 1: 1070, plates 5c, 7.2d, fig. 17. 2007.
Shrubs or trees, 20–50 dm, slender unless in full sun. Stems: twigs: new growth color not recorded, sparsely pubescent young, glabrescent, 1-year old deep reddish or deep purple-brown, 2-years old mid brown; thorns on twigs moderate to very frequent, straight to recurved, 1-year old bright, shiny, dark redbrown, slender, 2.5–4 (–5) cm. Leaves: petiole length 25–30% blade, sessile-glandular, glands few to several, small, adaxial sulcus pubescent; blade broadly elliptic to ovate, 4–6 cm mature, base cuneate to broadly cuneate or rounded, lobes 3 or 4 per side, max LII 15–25%, lobe apex acute, margins serrulate or crenate-serrate, veins 5–8 per side, slightly impressed, apex acute, abaxial surface glabrous young, vein-axils sometimes with axillary tufts, adaxial sparsely to moderately scabrous young, glabrate to sparsely scabrous mature. Inflorescences 5–15-flowered; branches sparsely to moderately pilose; bracteoles few, even young, light-brown, very narrow, margins glandular. Flowers 16 mm diam.; hypanthium glabrous or ± pilose proximally; sepals green with pale edges, triangular, 4 mm, abaxial surface glabrous, adaxial sparsely pilose; anthers pale-pink to pink; styles 4. Pomes red, becoming bright to deep red, ellipsoid to suborbicular, 8–10 (–12) mm, glabrous or sparsely pubescent; sepals spreading-reflexed to recurved triangular, glandular-serrate; pyrenes 3–5, dorsally grooved, sides ± plane to shallowly and smoothly concave concavity deepest on narrowest pyrenes.
Phenology: Flowering late May–early Jun; fruiting Aug–Sep.
Habitat: Thickets, light shade of aspen
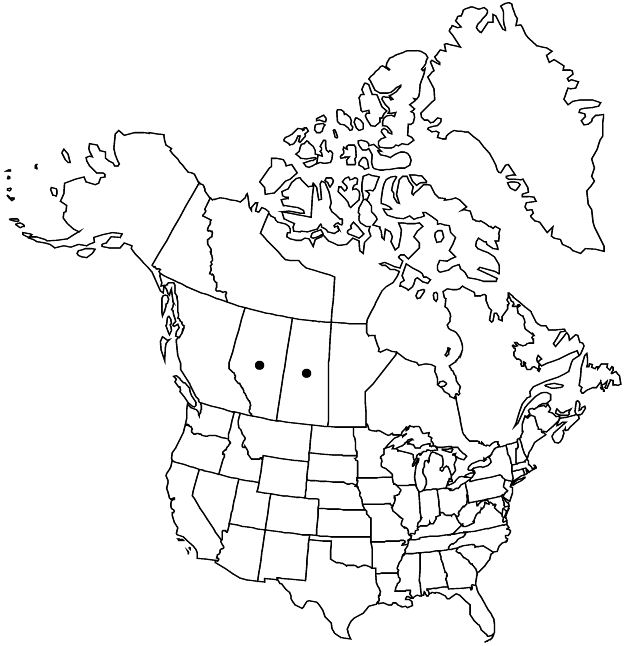
Elevation: 800–1200 m
Discussion
Crataegus rivulopugnensis, known only from the Cypress Hills, is widely distributed in the western part. The species is most similar to C. purpurella, though the color of the ripe fruit in the two species is quite different (red versus deep purple). They differ also in an almost allopatric distribution, habit when open-grown, and leaf size and numbers of veins per side (4 or 5 versus 3 or 4). Both species have less hairy adaxial leaf surfaces than most other sympatric hawthorns, often being glabrate adaxially at maturity.
INTERSERIAL HYBRIDS AND SPECIES OF HYBRID ORIGIN
Numerous putative interserial hybrids among North American hawthorns have been given binomials (J. B. Phipps 2005). Nearly all the named putative hybrids have proven to be rare or even apparently extinct. All of the 16 treated here satisfy at least two of the following criteria: there is a high degree of confidence that they are of interserial hybrid origin; they do not fit into any accepted series; they continue to be found long after their initial description, indicating either apomictic perpetuation, repeated creation, or both; or, they have appeared regularly as species in the literature, notably in works by E. J. Palmer. It is notable that most remain uncommon, suggesting that these are not particularly fit biotypes. Nevertheless, persisting populations of some (for example, Crataegus persimilis) may be readily found. The fifteenth example (C. turnerorum) is a well-collected, obviously persistent, local species in Texas.
The putative interserial hybrid, Crataegus florifera, however, is treated in the main text (in this case under ser. Anomalae) because it fits the serial description adequately and has significant and continuous ranges. Other examples included in the main text are some putative ser. Punctatae × ser. Crus-galli crosses that will be found associated with C. collina. The best studied interserial hybrids are two without names (though this will change with a forthcoming paper by the Dickinson group), both hybrids of the introduced C. monogyna. Here they are treated in the main text with their native parental species (C. punctata and C. suksdorfii). Neither of these hybrids is considered especially rare, and both appear to be increasing in frequency but so far there is no evidence of a fixed hybrid establishing.
Hybrids among North American native species have probably arisen multiple times (J. B. Phipps 2005); however, there was little evidence for the creation of successful new biotypes until some recent papers by the Dickinson group demonstrating modern derivatives of ancient alloploid of ser. Crataegus (E. Y. Y. Lo et al. 2009) and perhaps more recent alloploid derivatives from ser. Douglasianae (T. A. Dickinson, pers. comm.). Hybridization among more closely related apomicts may be more frequent than previously thought (Dickinson et al. 2008) but, with more cryptic characteristics, may more easily escape detection.
Selected References
None.
Lower Taxa
"thin" is not a number."sessile-glandular" is not a number."adnate" is not a number.