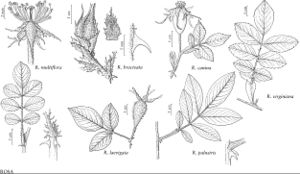
Rosa multiflora
in J. A. Murray, Syst. Veg. ed. 14, 474. 1784.
Stems erect, arching, procumbent, or climbing, 15–30 (–100) dm; bark of canes green to reddish-brown; infrastipular prickles paired, curved, rarely erect, stout, 4–6 × 3 mm, internodal prickles fewer, similar, aciculi absent. Leaves deciduous, 5–12 cm; stipules narrowly lanceolate, 8–13 × 1.5–2 mm, auricles flared, 4–8 mm, margins fimbriate, cleft to 3 mm, usually stipitate-glandular or gland-tipped, surfaces glabrous, sometimes puberulent, eglandular or sessile or stipitate-glandular; petiole and rachis with pricklets, puberulent, pubescent, or tomentose, usually stipitate-glandular, sometimes eglandular; leaflets (5–) 7–9 (–11), terminal: petiolule 7–13 mm, blade obovate to elliptic, 10–45 × 8–25 mm, membranous, base cuneate, margins 1 (–2) -serrate, teeth 12–20 per side, sharp, rarely gland-tipped, apex acute to long-acuminate, abaxial surfaces glabrous or pubescent (especially midveins), eglandular, adaxial green, dull, glabrous. Panicles 5–30+-flowered. Pedicels 5–12 mm, pubescent (especially proximally), sometimes glabrous, stipitate-glandular or eglandular; bracts 1 or 2, narrowly lanceolate, 5–10 × 1–2 mm, margins short-stipitate-glandular, surfaces glabrous, eglandular. Flowers sweet-scented, 1.5–2.5 cm diam.; hypanthium oblong, 2 × 1–1.5 mm, eglandular or stipitate-glandular; sepals ovate-acuminate, 6–10 × 1.5–2 mm shorter than petals, margins pinnatifid, tip 1–2 × 0.5 mm, abaxial surfaces glabrous, usually stipitate-glandular; petals single or double, white, rarely pale-pink, 7–13 × 5–9 mm; carpels 6–11, styles glabrous, exsert 3–4 mm beyond stylar orifice rims (0.5–1 mm diam.), hypanthial disc 2–3 mm diam. Hips orange-red to red, ovoid to globose, (4–) 5–7 (–10) × 5–7 mm, eglandular or sparsely stipitate-glandular. Achenes 6–11, tan, 3.8–5 × 2–2.5 mm. 2n = 14.
Phenology: Flowering Apr–Jun.
Habitat: Disturbed areas, thickets, borders of woods, fences, streamsides, roadsides, old pastures, fields, acidic soil
Elevation: 0–1700 m
Distribution
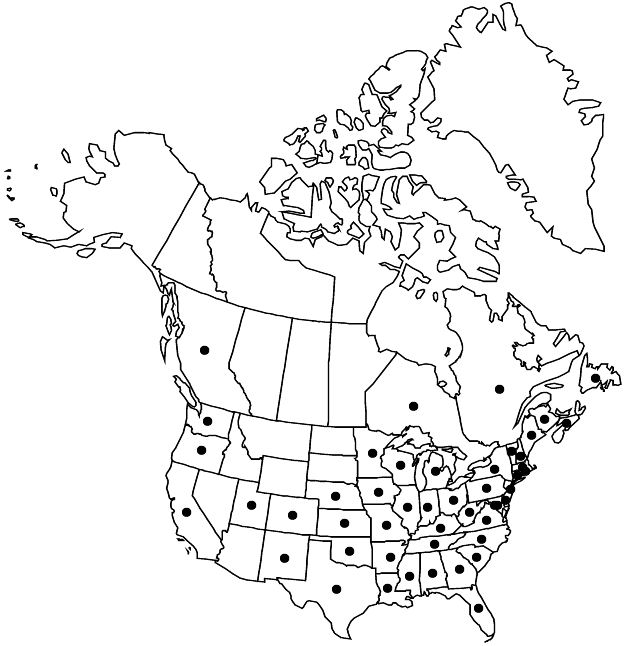
Introduced; B.C., N.B., Nfld. and Labr. (Nfld.), N.S., Ont., Que., Ala., Ark., Calif., Colo., Conn., Del., D.C., Fla., Ga., Ill., Ind., Iowa, Kans., Ky., La., Maine, Md., Mass., Mich., Minn., Miss., Mo., Nebr., N.H., N.J., N.Mex., N.Y., N.C., Ohio, Okla., Oreg., Pa., R.I., S.C., Tenn., Tex., Utah, Vt., Va., Wash., W.Va., Wis., Asia (China), Asia (Japan), also in Mexico, Pacific Islands (Philippines)
Discussion
Rosa multiflora was introduced in North America before 1811 as an ornamental. In the twentieth century, seedlings were widely provided by the U.S. Soil Conservation Service for growing hedgerows and similar plantings where it thrived in more acidic soil east of the Rocky Mountains. The species invaded pastures, degraded forage quality, reduced grazing area and agricultural productivity, and was widely reported as a noxious weed, causing, for instance, severe eye and skin irritations in cattle. Domestic sheep and goats feed on leaves, new buds, and shoots, and goats in pastures with severe R. multiflora infestations can virtually eliminate this rose within four seasons. The USDA Forest Service reported in 2006 that in 31 states the species had become a major conservation issue (S. E. Banasiak and S. J. Meiners 2009).
Vigorous climbers known as the “hard-luck” roses because of their great tolerance to frost, neglect, and varying habitats have been developed from Rosa multiflora, which has long served as a major root stock for budding roses in the rose industry worldwide.
Achenes of Rosa multiflora have long been used as a purgative in Japanese traditional medicine.
Selected References
None.
Lower Taxa
"dm" is not declared as a valid unit of measurement for this property."dm" is not declared as a valid unit of measurement for this property."dm" is not declared as a valid unit of measurement for this property.